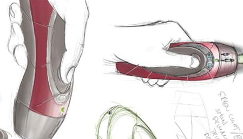
Injector gadget
Fear of needles and injections can be a prickly subject for many, however medical product design specialist Team Consulting has the answer.
Team has developed a design for a needleless injection system for use where mass vaccination is required. Not only does this remove the fear factor, but it also prevents contraction and spread of infectious diseases in animals or humans.

Team’s needless injection system
The design uses an ingenious method to deliver its load through the skin. “A mini combustion engine ignites butane in a compartment, which creates a very small, discrete and silent ‘explosion’,” explains Head of industrial design, Paul Greenhalgh. “The expansion of this gas forces carefully controlled amounts of medicine into a jet with sufficient pressure to pass through a patient’s skin.
Development of the product began with traditional sketching, but soon became more technical. Mathematical modelling in MathCAD was used to predict combustion performance and computational fluid dynamics to model the fluid dynamics for the ‘shot’.
Team uses SolidWorks as its principal engineering CAD tool, in conjunction with a Wacom Cintiq digital sketch pad and Adobe Illustrator for the industrial design.
The SolidWorks spline feature enabled curves to be traced and modified to create the geometry, while surface analysis was used to eliminate errors before a screenshot from SolidWorks was taken back into Illustrator to experiment with features, such as controls and displays.
The whole SolidWorks package was utilised: PDMWorks Workgroup product data management software enabled concurrent multidisciplinary working without the introduction of errors while SolidWorks PhotoWorks created photorealistic renderings.
It didn’t stop there. Functional components were machined directly from SolidWorks models and drawings. “SolidWorks multibody functionality was used to break the device into separate components which were sent to model making partners for the generation of rapid-prototyped, highly finished handling models for user trials,” adds Greenhalgh.
Working within a reliable workflow helps speed up Team’s design process – achieving good results in a rapid time frame needs experienced designers and a design solution that can be ‘life saving’.
www.team-consulting.com
Hear me now!
Most hearing aids work by amplifying sound through the ear canal and middle ear, but a new permanent solution uses bone instead of air to conduct sound.
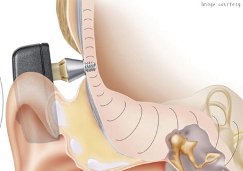
Cochlear’s Baha is attached directly to the bone, a sound processor amplifies sounds, transferring this directly to the inner ear
Currently around 55,000 people worldwide are using Cochlear’s Baha, an implantable bone conduction hearing solution. Attached directly to the bone, a sound processor amplifies sound, transferring this directly to the inner ear.
Cochlear claims that its solution provides a significantly better quality of hearing than most conventional, air conduction methods. A bone conduction sound processor distinguishes sounds so that sounds such as birds’ chirps and ocean waves can be magnified at the appropriate level.
It takes only one hour to place the implant behind the ear with the use of a local anaesthetic, and as the actual sound processor is connected to the implant, it can be removed with a simple press of a button, allowing the user to bathe, shower or swim.

The implant is placed behind the ear with use of local anaesthetic
The benefits of 3D CAD in the design of such a product are clear when you take into account its scale.
“We work with parts in hundreds and thousandths of a millimetre,” says Daniel Rådberg, senior design engineer and CAD manager, Cochlear. “It was a challenge to view these parts with the previous software programs we used, but in 3D it works perfectly.”
“Unlike those who produce and assemble our equipment, we do not work under a microscope to see the details,” he says. “However, we do have the ability to digitally magnify what we are working on. For us, the challenge is in managing small tolerances.”
The design team uses Autodesk Inventor amongst a range of specialised software from other industries. Daniel explains: “By working with both mobile telephony and dental implants, we understand how technology from very different worlds can work in the context of hearing aids.”
Cochlear’s motto is “Hear now and always”, which is appropriate for a technology that, due to its advances in design, is steadily growing in acceptance amongst the hard of hearing.
www.cochlear.com
Back to basics
Degeneration of the intervertebral disc, often called ‘degenerative disc disease’ (DDD) is a normal part of ageing, but for many it can cause severe and constant pain.
Acting as shock absorbers to prevent bone rubbing on bone, each disc has a tough outer, and soft water-based inner. The older one gets, the less water they store, and the more susceptible one becomes to injury.
Designed as a long term total disc replacement, the Compliant Artificial disc – Lumbar (CAdisc-L) produced by Ranier Technology benefits from controlled manufacturing technology to provide a durable elastomeric disc replacement. Having no fixed centre of rotation and with a degree of axial compliance the implant mimics the natural disc.
The device is comprised of polyurethane polycarbonate polymer with regions of differing modulus. A central relatively soft nucleus is encased by a region of progressively stiffer material, with the parts in contact to the vertebrae protected by hard end plates.
The CAdisc-L is designed using CT scan data from a library of past patients, with the geometry developed into a CAD model that allows further developments such as fixation points and surface features to be added.

The CAdisc-L is designed using CT scan data from a library of past patients, with the geometry developed into a CAD model that allows further developments
“Throughout the process, finite element modelling was used to optimise the design; to reduce internal stresses and to evaluate the flexibility of the proposed elastomeric core and material combinations,” says Ranier technical director Robert Snell, adding that the same COMSOL Mulitphysics analysis software was also used for CFD analysis to develop the mould filling and to optimise the material distribution.
The design team at the medical specialist firm primarily uses Pro/Engineer to help with the device and mould design, as well as the various connected instrumentation.
“One of the biggest design challenges was to ensure that the instrument design and device design remained in parallel,” explains Robert. “Keeping tight control on design versioning to ensure that the impact of any changes was rolled out through all design areas.”
The end result is only small, but it will come as a big relief to some of the millions of lower-back pain sufferers around the world.
www.ranier.co.uk
Stephen Holmes, gets his freshly sanitized hands on the latest in medical technology